Comment to ACIP: Need for Alignment Between ACIP and USPSTF Safety and Evidence Standards in Preventive Medicine
Deadline Midnight, 11.24.2025- "The agenda will include discussions on vaccine safety, the childhood and adolescent immunization schedule, and hepatitis B vaccines."
Link to comment: https://www.regulations.gov/document/CDC-2025-0783-0001/comment Please do so.
“Matters to be Considered: The agenda will include discussions on vaccine safety, the childhood and adolescent immunization schedule, and hepatitis B vaccines. The agenda will include updates on ACIP workgroups. Recommendation votes may be scheduled for hepatitis B vaccines. Vaccines for Children (VFC) votes may be scheduled for hepatitis B vaccines. Agenda items are subject to change as priorities dictate. For more information on the meeting agenda, visit https://www.cdc.gov/acip/index.html ”
To: Advisory Committee on Immunization Practices (ACIP)
Subject: Need for Alignment Between ACIP and USPSTF Safety and Evidence Standards in Preventive Medicine
Submitted by: Karl Kanthak
Date: 11.24.2025
Summary
I urge ACIP to adopt analytic and ethical standards equivalent to those used by the United States Preventive Services Task Force (USPSTF). Preventive interventions—administered to healthy individuals—must meet the highest possible safety threshold. Current ACIP practices apply therapeutic‑tier tolerance levels to vaccines, the quintessential preventive product. This inversion risks undermining public confidence by prioritizing administrative expedience over evidentiary integrity.
1. Failure to Apply Preventive‑Level Safety Standards
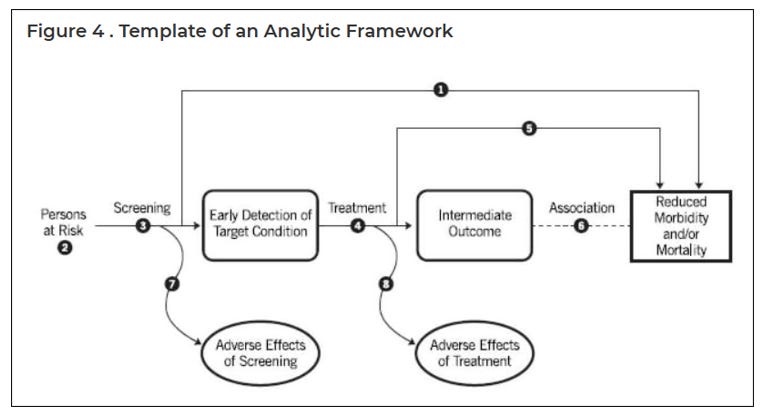
USPSTF’s framework interrogates both benefit and harm at each step of the preventive process, incorporating explicit feedback loops for adverse effects of screening and treatment. Its structure ensures continual, transparent self‑correction.
ACIP’s process, however, remains linear and closed—Manufacturer → FDA → ACIP → Population—with little systematic capacity for post‑market correction. Databases such as VAERS and VSD are either inaccessible or dismissed as non‑probative, leaving the committee without a functioning feedback mechanism.
Public analysis, “How the ACIP Deviates from the USPSTF Guidelines”, (Karl Kanthak, Substack, November 2025): https://karlkanthak.substack.com/p/how-the-acip-deviates-from-the-uspstf
2. Unquantified Risk in Simultaneous Administration
As detailed in my public analysis, “With Rare Exceptions All Vaccines Can Be Given at the Same Visit” (Karl Kanthak, Substack, December 2024):
The CDC currently permits co‑administration of nearly all live and inactivated vaccines, often at multiple anatomical sites during a single visit. While this guidance is operationally convenient, it implicitly assumes that all possible combinations and timing sequences have been tested for synergistic and cumulative safety. That assumption is empirically unsupportable.
According to FDA data, there are 109 licensed vaccines in the United States, including 16 routine vaccines and 6 combination vaccines, many with multiple formulations. The number of untested dosing permutations grows exponentially. No trial matrix exists—or could feasibly exist—that captures the safety profile of every concurrent combination across all ages and comorbidities.
Yet ACIP’s recommendations endorse such practice categorically, with no quantitative error margins or interaction modeling.
This contradicts preventive ethics: the less a harm is measurable, the smaller the population exposure should be—not larger.
3. Analytic Disparity and Consequences
By treating vaccination as an infallible preventive rather than an evolving intervention, ACIP insulates itself from the very scrutiny USPSTF mandates. The result is an overlapping inoculation regime in which biological products are administered simultaneously without comprehensive cross‑reactivity analysis.
Even basic contingencies—defective lots, mix‑ups, synergistic reactions—are treated as procedural exceptions instead of variables within policy modeling.
Public trust depends on honesty about uncertainty, not its omission.
4. Recommendations for Structural Reform
To realign ACIP with established preventive standards, the following reforms are warranted:
Adopt a USPSTF‑style analytic template with explicit harm‑benefit feedback loops.
Require empirical evaluation of concurrent vaccine administration using factorial or systems‑pharmacology modeling before categorical approval.
Establish open data access to anonymized VAERS and VSD records for independent analysis.
Incorporate outcomes data from natural infection recovery into population‑wide health metrics. Studies from Japan and Europe suggest that recovery from febrile childhood illnesses correlates with reduced lifetime cancer and autoimmune risk.
Publish cumulative risk reports quantifying product‑interaction uncertainty so the public can weigh knowns and unknowns transparently.
5. Conclusion
Preventive medicine’s legitimacy depends on stricter—not looser—standards of safety. Vaccines remain essential tools, but science is not weakened by transparency; it is strengthened by it.
ACIP can restore trust and reassert its scientific authority by acknowledging uncertainty, recalibrating risk tolerance, and aligning its methodology with USPSTF’s evidence‑based framework.
Respectfully submitted,
Karl Kanthak

Excellent testimony as always, Karl. Thank you!
Eloquent and important